Why post-event lead nurturing is different in Life Sciences
Time and resources are limited, which means the ideal lead-nurturing strategy is rarely realistic. In recent posts, we’ve broken down the causes and consequences of ineffective event lead nurturing. The unavoidable question now is what can actually be achieved in practice, given real constraints.
As science marketers, we often talk about why ROI underperforms, look at industry trend data, and debate which tools or lead capture methods to use. But all of that dodges the harder, more important question: how do you actually nurture leads over time in a way that feels credible to the audience and still delivers revenue?
For science marketing leaders already investing six figures in exhibition space, the challenge isn’t in the volume of data being collected; but in the reality of having to sustain momentum across long buying cycles. How do you move beyond the ‘thank you’ email sent after the trade show, when you’re already stretched thin?
In 2009, Stefan Stremersch and Wouter Van Dyck published the seminal paper Marketing of the Life Sciences. Its arguments were later distilled by IESE Business School in A Prescription for Marketing Medicine [1], which explored why Life Sciences marketing operates under fundamentally different rules. Central to this is the idea of “scientific linkage”: the recognition that physicians (or Principal Investigators (PIs) in our context) act as expert agents on behalf of end users, and that influence is exercised across distinct decision points throughout the product development and marketing lifecycle.

This isn’t an academic detour. The relevance for marketers is straightforward: in Life Sciences, compliance is not a constraint on trust, it is the foundation of it. The long buying cycles that frustrate commercial teams are not inefficiencies to be engineered away; they are deliberate safeguards. Organisations maintain these barriers until they are confident in the scientific, regulatory, and operational impact of a solution on their workflows. As science marketers, our role is to supply a defensible evidence trail that allows multi-stakeholder committees to lower those barriers with confidence.
Before moving into checklists and timelines, one principle matters more than all others. Trust in Life Sciences is not built through volume; it is built through intentional strategy over time. With limited time and resources, effective execution depends on finding the path of least resistance to scientific credibility. As you work through the next sections, anchor your approach to these three implementation principles:
- Repurpose, don’t invent
You almost certainly already have the evidence: posters, abstracts, validation data, technical sheets. The work is not creating more content, but packaging existing proof for the right stakeholder at the right moment.
- Focus on the expert agent
If you only have the capacity to nurture one individual, nurture the person who must justify your solution to everyone else. That internal advocate carries disproportionate influence.
- Watch for collective intent
One click signals curiosity, but multiple stakeholders from the same lab engaging points to a live project. Optimise for the project, not the individual lead.
The post-event window (90 days): from recall to readiness
If you’re process-minded, your instinct here is to build sequences. Resist that. In a buying cycle this long, sequences create noise; what you need instead are deliberate pulses. The goal in this window is a high-value, low-frequency presence that respects the protective buying behaviours we’ve already discussed, while positioning you as a credible guide rather than a persistent vendor.
The prospects who are genuinely ready to buy are already far down the path. They tend to make themselves obvious. You know how to spot them, sales knows how to spot them, and we’ve covered that dynamic elsewhere. This phase is not about accelerating the obvious deals. It’s about everyone else: the majority who are evaluating, socialising risk internally, and deciding whether you’re trustworthy enough to stay in the conversation at all.
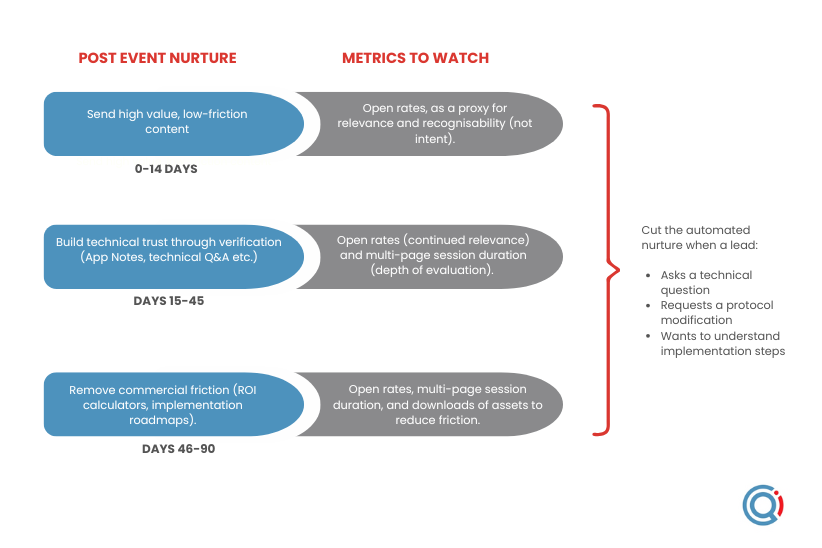
Phase 1: The memory anchor (the first 14 days)
This is where ‘scientific linkage’ moves from theory to leverage. Your brand has achieved recognition, the task now is to build enough trust to engage the buying committee beyond its protective barrier, starting with the person in that group who might be able to validate your credibility internally.
- Overall strategy: Building technical trust through verification not persuasion
- Tactic: Use secondary evidence such as third-party papers, technical Q&A, application notes to reinforce the original memory anchor.
- The goal and anchor: Replace early positive sentiment with defensible confidence by answering questions of technical fit, regulatory alignment, and practical compatibility. the outcome you want is not “this looks interesting,” but “this stands up to scrutiny.”
- Metrics to watch: Open rates (continued relevance) and multi-page session duration (depth of evaluation).
Phase 2: technical validation (days 15-45)
This is where ‘scientific linkage’ moves from theory to leverage. Your brand has achieved recognition, and the next step is to build enough trust to engage the buying committee beyond its protective barrier, starting with the person in that group who can validate your credibility internally.
- Overall strategy: Building technical trust through validation not persuasion.
- Tactic: Use secondary evidence such as third-party papers, technical Q&A, and application notes to reinforce the original memory anchor.
- The goal and anchor: Replace early positive sentiment with defensible confidence by answering questions of technical fit, regulatory alignment, and practical compatibility. The outcome you want is not “this looks interesting,” but “this stands up to scrutiny.”
- Metrics to watch: Open rates (continued relevance) and multi-page session duration (depth of evaluation).
Phase 3: The momentum (days 46-90)
This stage is about business, not science. The technical case has already been made. What you’re confronting now is behavioural friction: status quo bias and loss aversion. Research cited in The Jolt Effect by Matthew Dixon and Ted McKenna shows that 40–60% of deals where buyers express clear intent end in no decision at all [2]. Not a loss. Not a win. Nothing.
The underlying reason is well established in behavioural science. Humans are wired to prefer an imperfect status quo over the perceived risk of loss, a dynamic explained by Daniel Kahneman and Amos Tversky, whose work underpins much of The Jolt Effect. The practical implication for marketers is simple: at this point, you are no longer competing against another vendor. You are competing against inaction.
- Overall strategy: Identifying an active buying project and remove commercial friction.
- Tactic: Assets that make change feel safe and operationally contained: ROI calculators, implementation roadmaps, clear transition narratives (“here’s how straightforward this was for x organisation”), and consultation offers triggered only when warming signals are present.
- The goal and anchor: Reframe the decision so that staying with the current approach feels harder, riskier, or more costly than moving forward
- Metrics to watch: Open rates, multi-page session duration, and downloads of your assets designed for the high friction phase (calculators, roadmaps, implementation content).
The final metric to look out for here is the ‘heat’ of group intent, seeing how many people in that account are downloading the same high-value resources.
Role aware nurturing: defining the ‘who’
The Life Sciences industry spans hundreds, often thousands, of distinct job titles. Even when narrowed to core decision-making groups, the complexity is still too high to design individual nurture paths without burning time and resources you don’t have.
The goal here is not perfect personalisation; it’s effective alignment. You need a simple framework that ensures each decision group receives the type of evidence they require, while allowing you to operate efficiently at scale. That’s where role-aware nurturing comes in.
Use the matrix below to categorise your leads by decision role rather than job title. Then assess how your follow-up aligns with the evidence each group needs to progress. When done correctly, this approach preserves credibility with the buyer and protects your team’s capacity at the same time.
| Stakeholder role | Their goal / filter | The ‘evidence’ they need |
| The technical validator (Bench scientist/Post-Doc) | Reproducibility: Will this protocol work in my lab day-to-day? | Raw data, methodology, application notes. |
| The expert agent (Principal Investigator/Lab Head) | Strategic impact: Will this advance my research or grant potential? | Peer-reviewed citations, innovation roadmaps. |
| The internal influencer (Core Facility Manager/Bioinformatician) | Compatibility: How does this fit into our existing infrastructure? | Integration specs, software compatibility, data output formats. |
| The non-decision maker (Lab Manager/Admin) | Operations: Is the onboarding and maintenance seamless? | Service-level agreements (SLAs), training schedules, logistics. |
| The risk mitigator (Procurement/QC Manager /Finance) | Viability: Is the investment safe and compliant? | GMP/ISO certifications, ROI calculators, “Total Cost of Ownership” (TCO). |
By looking at it this way, you’re not building a nurturing path for thousands of guessed job titles, you’re building a nurturing path for their goals.

Re-using event assets: content sustainability vs smart asset management
When time, budget and headcount are limited; reuse of content goes a long way. Most teams already have a lot more useable content than they realise; the failure is not scarcity but it’s poor structuring. Let’s make a simple assumption: you have a scientific poster, a technical presentation and a live product demo. Across the 90-day post-event window we discussed, those three assets can realistically generate at least nine distinct, purpose-built touchpoints.
This framework isn’t meant to capture every concern of every buyer. It’s a snapshot that demonstrates how event content can be systematically reused without diluting credibility or overloading your team.
Think in stages, not formats.
Stage 1: memory anchor
The objective is recognition, not depth. Use small, high-impact snapshots: a single headline graph from the poster, a screenshot of a key data point from the presentation, or a short highlight clip from the demo.
Stage 2: trust development
Now you earn legitimacy. This is where you expand slightly: a materials-and-methods PDF derived from the poster, a two-minute narrated walkthrough of the technical slides, or a concise spec sheet extracted from the demo.
Stage 3: momentum and validation
At this point, content must support internal justification. Arm the PI with citations or third-party papers linked to the poster, provide a strategic impact brief that reframes the technical presentation in operational terms, or deliver a lab-readiness checklist based on the demo.
Execution reality: living lean
Everything discussed in this article lives in the real marketing world, not in an idealised automation diagram. Yes, we have tools that automate, score, and signal intent, but those tools are still operated by people with finite time, attention, and energy. The point of this framework is not to prescribe perfection. It’s to show the rationale, outline the ideal, and then deal honestly with what’s achievable.
A common fear is that the only way to “do this properly” is with an expensive, fully integrated tech stack: a 12-month automation journey, thousands of tailored paths, adaptive logic across multiple buying groups, all running on a seven-figure platform. That belief is paralysing; and wrong.
You do not need perfection to make progress.
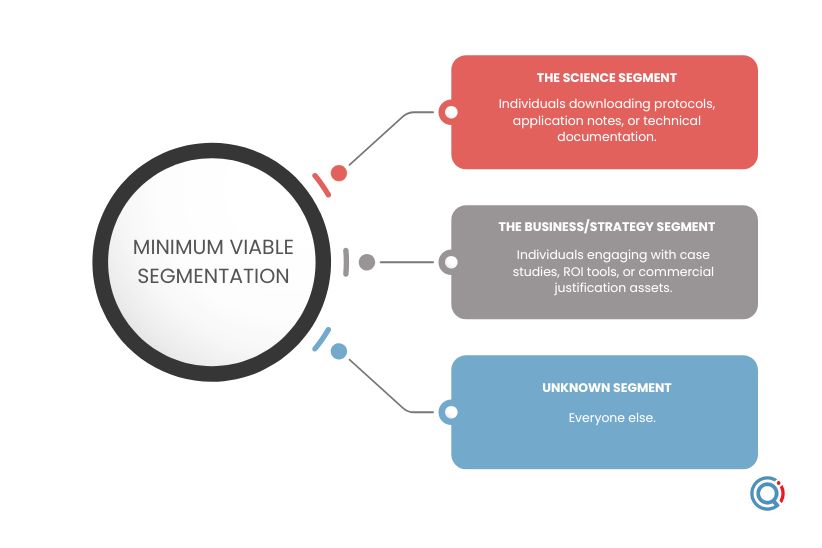
With limited time, limited resources, and partial or non-existent automation, you can still apply minimum viable segmentation. This is a proven approach that focuses effort on a narrow, high-impact beachhead: a small group of prospects with similar needs that can be reached efficiently with a relevant message.
In practice, this can be reduced to three segments:
- The science segment
Anyone downloading protocols, application notes, or technical documentation.
- The strategy segment
Anyone engaging with case studies, ROI tools, or commercial justification assets.
- The unknown
This is everyone else.
If you can’t do anything more sophisticated than this, do this. It will outperform most overengineered journeys that never get executed properly.
Where automation actually helps
Automation is useful when it removes manual effort, not when it becomes the strategy itself. Used correctly, it supports delivery of the right evidence across the ninety-day post-event window without exhausting the team.
In practical terms, automation can serve three roles:
- Triage
Tagging and routing leads based on engagement helps distinguish bench scientists from Principal Investigators (PIs) and commercial stakeholders without guesswork.
- Early warning
Rules based on high-intent behaviour, such as time spent on late-stage assets, surface accounts that warrant human follow-up.
- Maintaining a steady marketing ‘heartbeat’
Not all leads move forward in a straight line. Automation can maintain a low-frequency presence and shift contacts into different nurture paths when engagement drops, without overwhelming either the buyer or the team.
When automation hands over to a human
Return to the shield for a moment. People matter at the point where it starts to lower. The moment a lead asks a technical question, requests a protocol modification, or wants to understand practical implementation steps, automation has done its job. At that point, cut it.
Automation gets you to the door, and then humans walk through it. The same logic applies to triage and early-warning signals; automation identifies opportunity and risk, then people create the momentum.
Measuring progress without forcing false ROI
If you measure success purely by closed-won deals in the first 90 days, every event will look like a failure. Similarly, looking simply at raw engagement volume leads is going to send you into the opposite and equally incorrect conclusion that your content is working. What we really want to measure is the velocity of trust.
This is where the age old adage still holds; quality beats quantity. A thousand views of an event highlight reel is great but vanity. However, 10 downloads of a technical integration spec from the same domain is an solid intent signal. So what other warming signals should we be looking for?
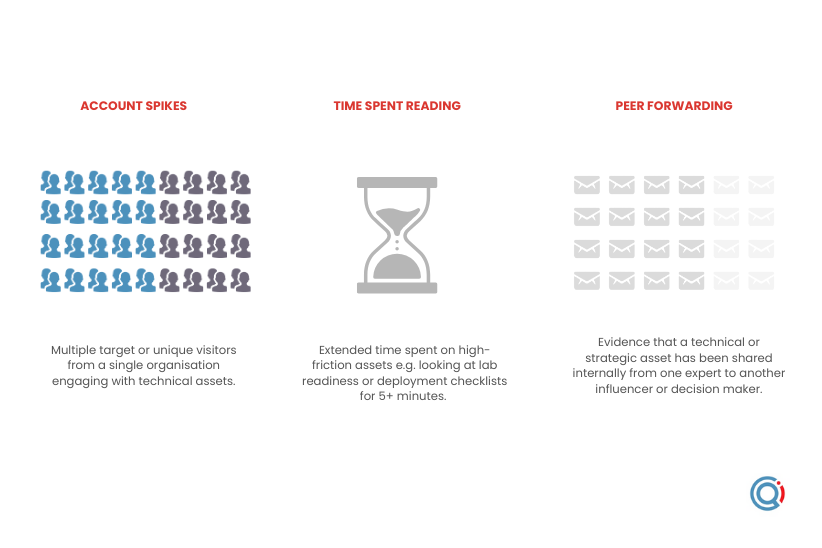
Warming signals to watch for
Account spikes: Multiple target or unique visitors from a single organisation engaging with technical assets.
Long term reading: Extended time spent on high friction assets e.g. looking at momentum stage (days 46-90) resources for 5 minutes.
Peer forwarding: Evidence that a technical or strategic asset has been shared internally from one expert to another influencer or decision maker.
When to pause and when to escalate
Escalate: When individual engagement turns into collective intent. That is your cue to move from automated nurturing to human outreach (and pass that information to your commercial folks for closing).
Pause: If a lead has not engaged with content from the first two phases, they are not ready to care about implementation details, ROI, or pricing. Move them to a low-frequency nurture that maintains the marketing heartbeat without applying decision pressure.
Final thoughts: A compass, not the play-by-play
This guide is not an end-to-end process ma, with an answer to every scenario and every buyer group. It is a framework designed to help you make sensible trade-offs when time, content and automation are constrained.
In an ideal world, we would have unlimited resources. Every prospect would sit in a precisely tailored nurture path, dynamically adapting across phases and eventually converting, regardless of how long it took. But that world does not exist, and chasing it is a distraction.
What does exist are a few fundamentals worth keeping front of mind:
1. Leads are not linear. Not every prospect moves cleanly from phase one to phase three. Some exit early. Some linger in awareness for months before progressing. Others stall at technical validation and never move beyond it. This is normal buyer behaviour, not a marketing failure.
2. Exiting the path is a valid outcome. It is neither commercially nor operationally sensible to keep every lead in a high-touch nurture cycle. When engagement drops, reallocate effort. Use automation and low-frequency follow-up to maintain presence without draining resources.
The objective throughout this framework is alignment, not volume. The goal is to build trust where it can be earned, not to maintain endless nurture paths for their own sake.
References
- Stremersch, S., & Van Dyck, W. (2009). Marketing of the life sciences. IESE Business School Insight. [https://www.iese.edu/insight/articles/prescription-marketing-medicine/]
- Dixon, M., & McKenna, T. (2022). The Jolt Effect: How High Performers Overcome Customer Indecision. Portfolio / Penguin Random House. [https://www.amazon.co.uk/Jolt-Effect-Performers-Overcome-Indecision/dp/0593538102]